Celebrating Motherhood as a Cancer Mom “On Mother’s Day I can think of no mother more deserving than a mother who had to give one back.” ~ Erma Bombeck Today we celebrate motherhood. For some it is a day of great joy and happiness. For others – those who have lost a child, those who are longing to become a …
2022 Financial Report
What an amazing 2022! We are incredibly grateful to the wonderful partners and benefactors nationwide that allow the Beat Childhood Cancer Foundation to provide this overview highlighting a year of transition, success, commitment, and hope. For more than a decade, the Beat Childhood Cancer Foundation has funded and built a precision medicine program aimed at finding better treatments by fueling …
The last day of September
September is Childhood Cancer Awareness Month; the ribbon color is gold. We spend all year working on driving bold new options for kids fighting cancer, through a research consortium we helped build, now in over 60 children’s hospitals and research institutions internationally. Here’s some ways you can support on the last day of Childhood Cancer Awareness Month in 2021!Into the …
Finding Meaning
One day can change our vision of the world. For me, that day was September 23, 2005. What I know now has enhanced my sense of purpose personally and professionally. #findingmeaning #hope #inspiration #knowing 15 years ago today, life changed forever. I thought I was aware, I thought I appreciated the fight, I thought I understood the challenges and realities …
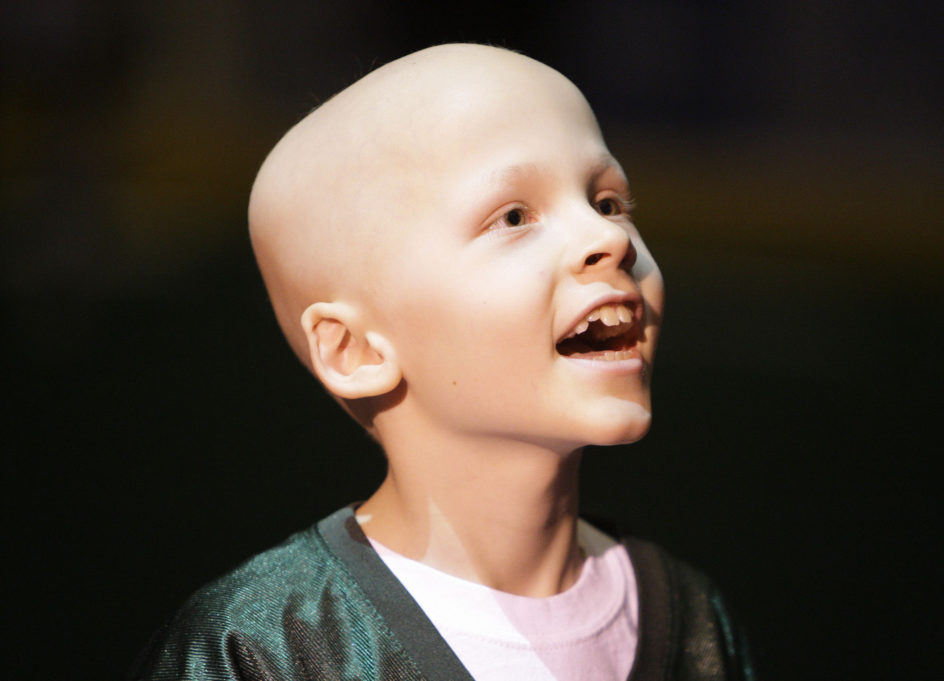
Growing Hope to Beat Childhood Cancer
“In this earth, in this soil, in this pure field, let’s not plant any seed other than seeds of compassion and love.” ~ Rumi The philanthropic efforts of the families, friends, communities, and team members that have contributed to the mission and vision of the childhood cancer endeavors my family has spearheaded, supported, and sustained have all been focused on …
2020 Vision: A Blog by Annie Bartosz
Are you Listening to me? How often in the last 18 years has every high school graduate heard those words? Parents, teachers, siblings, friends . . . the list goes on. But, all too often, we are too distracted to really listen. Hearing is the physical ability to detect noise; whereas, listening involves mental processing, taking in information and making …
The Luxury of Legacy
“If you’re going to live, leave a legacy. Make a mark on the world that can’t be erased.” Maya Angelou By simple definition, a legacy is a gift of money or property, a bequest, or something handed down upon death. Such definition seems a bit antiquated, however, as if a legacy can only be ‘things’ or ‘gifts’ of the past; …
What Is . . .
. . . hope? Social distancing can not and will not stop the spread of awareness, knowledge, and action. Walking around the neighborhood with my kids last night, I was delighted to see the letters H-O-P-E colorfully painted and hanging in a front window. We stopped to admire the thoughtful handiwork with a smile and, in the moment, truly felt …
Gratitude for the Great Gold Team
“Gratitude turns what we have into enough.” Aesop Gold In September was built on hopes that began within the hearts and home of my family; but the truth is that the definition of family has always extended far beyond a single kitchen table to include the family of supporters, donors, volunteers, team members, advocates, and leaders who have helped to …
The Greatness of Community
“The greatness of a community is most accurately measured by the compassionate actions of its members.” ~ Coretta Scott King What words can possibly be offered during these current days of fear, uncertainty, disbelief, and confusion? The simple answer: none. As someone who loves words and has used them to lift my spirit and heal my heart during the worst …